The Internet of Medical Things (IoMT) in the healthcare setting has moved past the experimentation stage and has become an active infrastructure. From surgery-assisting robotic arms to ingestible sensors, the medical Internet of Things is now embedded in how leading healthcare providers operate, monitor, and deliver care. And the market statistics confirm the trajectory.
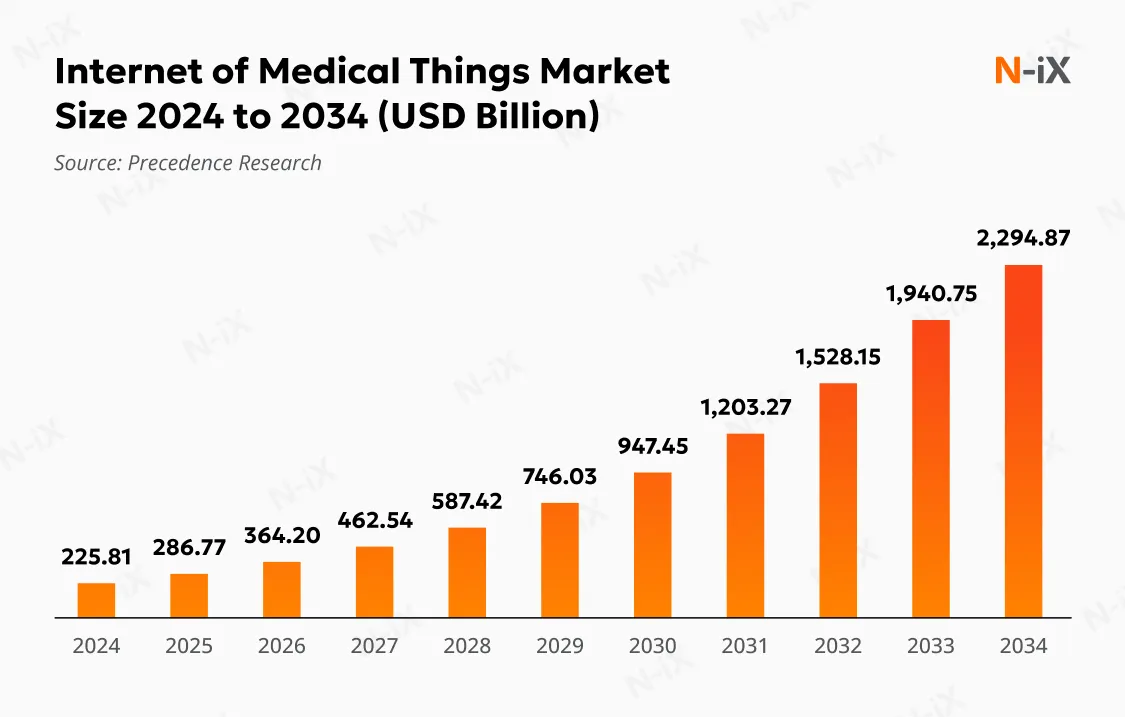
According to Precedence Research, the global IoMT market stood at $286.77B in 2025 and is projected to exceed $2,294.87B by 2034 [1]. In addition, as the direct-to-consumer (DTC) healthcare model gains popularity, IoT solutions are becoming a staple for those offering alternatives to conventional healthcare and seeking to tap into a rapidly growing market.
This article covers the major aspects of the Internet of Medical Things in healthcare software development, including its types, benefits, and how to address implementation challenges.
What is the Internet of Medical Things?
Internet of Medical Things, IoMT, is a network of connected medical devices and software applications that collect, exchange, and act on health data, often without manual input at every step.
IoMT devices capture clinical signals: vital signs, glucose levels, ECG readings, medication adherence, patient location. That data is transmitted to healthcare IT systems, such as EHRs, clinical dashboards, and hospital information systems, that care teams use in their practice.
The software layer matters as much as the hardware. IoMT applications analyze incoming device data to enable remote diagnostics, continuous monitoring, and personalized treatment at a scale no manual workflow can replicate.
IoMT devices are typically grouped by the environments in which they operate. Each category comes with its own integration requirements, regulatory considerations, and clinical use cases. Let's review them.
Types of IoMT devices
The Internet of Medical Things spans a wide range of hardware—from smart thermometers and infusion pumps to ingestible sensors and robotic surgical systems. Devices are typically grouped into five categories based on where they operate:
- In-home IoMT;
- Wearable IoMT;
- Mobile IoMT;
- Public IoMT;
- In-hospital IoMT.
Let's review each in more detail.

In-home IoMT
In-home medical Internet of Things devices are installed in patients' residences to support ongoing care without clinic visits. They cover remote monitoring, diagnostics, and medication management, making them particularly valuable for patients with chronic diseases and the elderly.
Common examples include smart pill dispensers, connected blood pressure monitors, glucometers, connected inhalers, and smart medical beds. Personal emergency response systems sit in this category, too. When a patient experiences an emergency at home, the device automatically alerts the care team, without the patient needing to initiate contact.
Wearable IoMT
Wearable or on-body Internet of Medical Things devices attach to the body and continuously transmit health data to remote monitoring systems. They fall into two distinct tiers.
Consumer-grade devices, such as fitness trackers, Apple Watches, and Fitbits, track wellness metrics for personal use. They don't require clinical supervision and can feed data into a provider's portal, but they are not certified medical instruments.
Medical-grade devices are a different category entirely. These are regulated, clinically validated tools used under professional guidance. FreeStyle Libre, Abbott's continuous glucose monitor (CGM), is a widely deployed example. It measures interstitial glucose in real time and transmits readings directly to the care team, eliminating routine finger-prick testing [2].
This distinction matters for procurement and compliance. Medical-grade wearables carry stricter certification requirements and integration obligations than consumer devices do.
Mobile IoMT
Mobile IoMT pairs a physical device with a smartphone application connected to a hospital system or provider portal. These tools use NFC, RFID, and Bluetooth to communicate with clinical systems on the move.
KardiaMobile by AliveCor is a practical example. The device records a medical-grade electrocardiogram (EKG) directly on a smartphone and transmits the data to a physician for review. It detects atrial fibrillation, bradycardia, and tachycardia, enabling continuous cardiac monitoring outside of a clinical setting [3]. With each membership tier, the device can detect even more conditions.
Public IoMT
Public Internet of Medical Things, also known as community IoMT, extends the connected care infrastructure into public spaces and underserved areas. The goal is straightforward: bring primary care access to populations that lack it.
A telehealth kiosk is a popular example. Equipped with video displays, HD cameras, and integrated medical instruments (blood pressure cuffs, glucose monitors, stethoscopes, etc.), these kiosks enable full remote consultations. Patient data feeds directly into the provider's system. REDYREF's telehealth kiosk is one of the operational models on the market [4].
For organizations evaluating rural or community outreach strategies, the public Internet of Medical Things should be included in the roadmap.
In-hospital IoMT
In-hospital IoMT connects the devices already used in the facility, such as infusion pumps, sensor-equipped beds, wheelchairs, and monitoring equipment, into a system that clinical staff can oversee from a central dashboard. Nurses monitor stable patients remotely and attend to critical patients in person. Alerts fire automatically on abnormal readings, decreasing the response time.
Discover how robotics is revolutionizing healthcare – access the comprehensive overview now!


Success!

The hospitals also use the Internet of healthcare things, a broader term for IoT devices for healthcare facilities, inventory, and asset management. Internet of things healthcare applications help organize and keep medical equipment in place, run device diagnostics, and facilitate equipment search. In addition, they can also be used to monitor hygiene and compliance with treatment protocol.
Explore further: IoT in healthcare: key benefits and use cases
Applications of IoMT in healthcare
Internet of Medical Things applications do two things: they capture clinical or operational data, and move that data to where it can drive a decision. The examples below show what that looks like in practice and what it means for patient outcomes and operational efficiency.
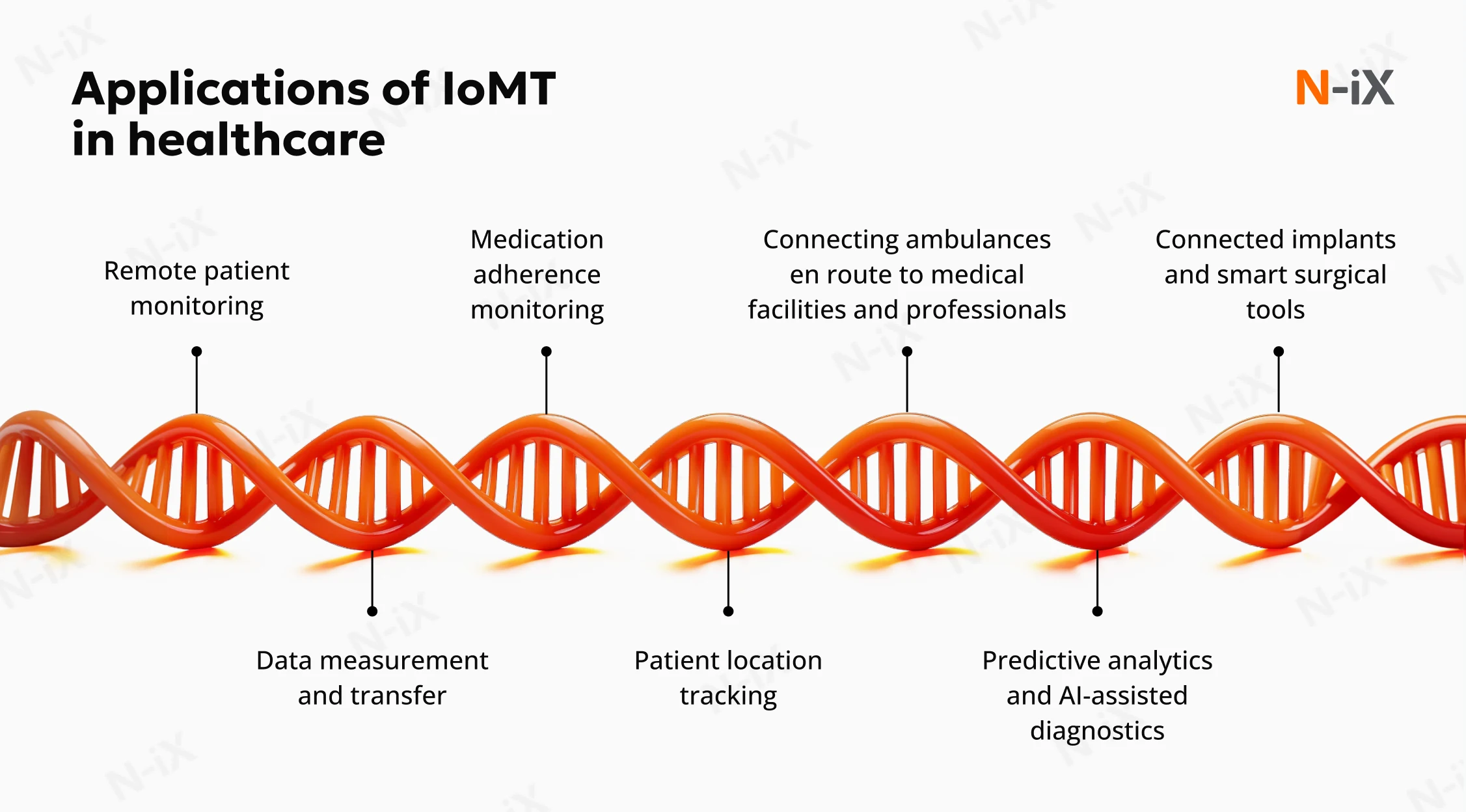
Remote patient monitoring
IoMT devices enable continuous remote patient monitoring (RPM) of vital signs, such as heart rate, blood pressure, glucose levels, oxygen saturation, without the patient leaving home. A diabetic patient using Dexcom G6, for example, transmits real-time glucose readings directly to their care team. The physician sees trends, adjusts medication, and intervenes without the patient scheduling an in-person visit.
IoMT-based RPM also allowed the hospital-at-home (H@H) model to gain traction. Mass General Brigham is actively shifting 10% of its medical patients to home-based care. This move is backed by CMS's Acute Hospital Care at Home initiative, which permits hospitals to deliver inpatient-level care outside their walls [5].
IoMT-enabled RPM works within hospital walls too. Nurses monitor stable patients remotely, optimize their visits, prioritizing the needs of critical patients while effectively monitoring those in the general wards. In a workforce shortage conditions, it prevents nurses from burnout while ensuring proper care for inpatients.
Data measurement and transfer
Medical IoT helps collect and transmit real-time health data to healthcare systems, enabling providers to monitor patient conditions. Devices such as wearable ECG monitors and smart blood pressure cuffs transmit data to physicians, enabling quick responses to abnormal readings or worsening health conditions.
Medication adherence monitoring
Non-adherence to medication is one of the most persistent problems in chronic disease management. Smart pill dispensers address it directly. They track whether doses are taken on schedule and send alerts to caregivers or providers when doses are missed. Mobile apps reinforce this with push notifications and reminders.
Patient location tracking
For hospitals treating elderly patients or those with dementia, Alzheimer's, or severe mental conditions, patient wandering is a real safety risk. Wearable GPS trackers alert staff the moment a patient leaves a designated area, enabling faster response and reducing incidents.
The more advanced Internet of Medical Things devices go further. On-device AI enables fall detection and real-time vital sign monitoring, providing caregivers not only with the patient's location but also whether the patient needs immediate help.
Connecting ambulances en route to medical facilities and professionals
IoMT helps align the work of ambulance crew and emergency departments. Vital signs, ECG readings, and preliminary diagnostics captured en route are transmitted to the hospital in real time. The receiving team knows what is coming before the patient arrives and can have the right room, personnel, and equipment ready.
Predictive analytics and AI-assisted diagnostics
Modern medical IoT solutions no longer just report what is happening. Combined with AI, they forecast what is about to happen.
Continuous data streams from connected devices feed predictive models that flag deterioration risks, forecast readmissions, and identify patients likely to decompensate before clinical signs are obvious. For technology leaders, this is the application that shifts IoMT from a monitoring tool to a clinical decision support system. It is also the area with the steepest integration requirements, addressed in the challenges section below.
Connected implants and smart surgical tools
Connected implants and surgical tools depend on IoMT infrastructure with sensor feeds, real-time data transmission, and integration with imaging and EHR systems to function.
Orthopedic implants that monitor healing in real time and smart surgical instruments that provide intraoperative feedback are now in active clinical use. For example, Medtronic's BlueSync technology connects pacemakers directly to a patient's smartphone via low-energy Bluetooth, transmitting cardiac data continuously and flagging anomalies without a clinic visit [6].
On the surgical side, AI-assisted robotic systems have demonstrated a 25% reduction in operative time and a 30% decrease in intraoperative complications compared to manual methods [7].
For organizations mapping their IoMT strategy, connected implants and smart surgical tools represent a high-complexity, high-impact frontier worth planning for now.
Explore how robotics can be used in healthcare
Benefits of IoMT for healthcare providers
While adopting IoMT is a clinical upgrade, it also becomes a business lever. It reduces the cost of delivering care, extends capacity without adding headcount, and positions providers to compete in a market that is shifting fast toward outcome-based reimbursement. Here is what the investment translates to in practice.
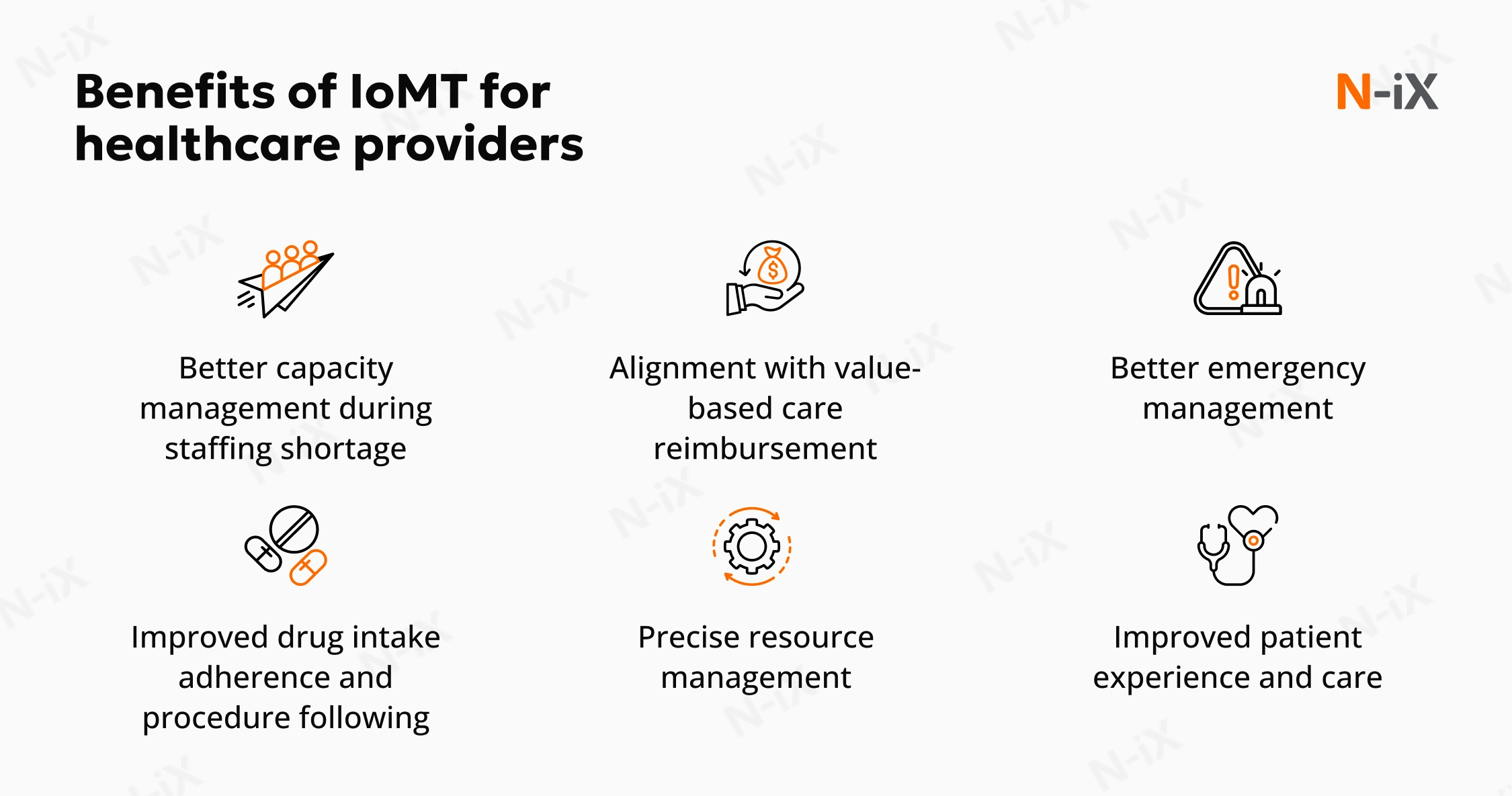
Better capacity management during staffing shortage
Medical Internet of Things devices enable continuous, remote monitoring of patient vitals, allowing nurses and physicians to oversee more patients without increasing the time spent on routine checks. Staff attention is redirected to where it is most clinically needed. In facilities operating at reduced capacity due to staffing maldistribution or shortage, workload optimization becomes a necessity.
Alignment with value-based care reimbursement
Under the fee-for-service model, there was limited financial incentive to invest in remote monitoring. The providers didn’t get paid for a hospital visit that didn’t happen. Under the value-based care (VBC) model, that prevented visit is exactly what earns revenue. Payers are rewarding outcomes, not volume, and infrastructure involving medical IoMT devices helps make outcomes measurable and manageable.
Care providers with continuous patient data, such as glucose trends, cardiac rhythms, and blood pressure patterns, can document the clinical picture that value-based contracts require. Those without it are operating on assumptions and may have little visibility into the patient's progress or outcomes.
Better emergency management
Internet of Medical Things improves emergency outcomes on two levels. First, it enables continuous remote monitoring of high-risk patients and triggers automated alerts when readings fall outside safe thresholds, catching deterioration before it becomes a crisis. Second, it connects ambulances to receiving facilities in real time, allowing hospital teams to prepare the right care environment before the patient arrives.
This matters most in time-critical conditions. In cardiac events, every minute of preparation time saved translates directly into higher survival probability. It also enables remote specialist consultation while the patient is still in transit. This directly affects patient outcomes, especially in facilities facing physician shortages or in rural settings with thin specialist coverage.
Improved drug intake adherence and procedure following
For providers managing patients with chronic diseases including diabetes, hypertension, COPD, and heart failure, medication non-adherence is both a clinical and a financial problem. Missed doses drive complications, readmissions, and avoidable downstream costs.
Dispensers and monitoring tools with integrated Internet of Medical Things close this gap. They track actual intake, alert providers when patients go off-plan, and give clinical teams the data to adjust dosages or switch medications based on real adherence. In geriatric care and memory disorder treatment, where self-management is unreliable, this visibility is paramount to successful patient treatment.
Precise resource management
Medical equipment that is missing when needed creates delays, safety risks, and operational friction. Real-time location systems (RTLS), enabled by IoMT technologies, give clinical staff live visibility of critical equipment (e.g., ventilators, defibrillators, infusion pumps) without requiring physical checks. Staff locations can be tracked and managed the same way, ensuring the right personnel reach the right unit without delay.
The same logic applies to emergency units. IoMT-enabled fleet management for ambulances and emergency vehicles allows operators to assign the closest available unit to an incoming call. In large healthcare facilities, IoMT can help monitor the location of medical staff, so the right personnel can be deployed to the correct urgent care units faster.
Improved patient experience and care
By gathering patient data promptly and precisely, medical devices powered with IoT technologies help doctors and hospital personnel to provide better treatment and care. In addition, it gives patients visibility of their conditions, fostering a better culture of self-care and awareness.
It's not only medical providers who can benefit from these technologies. The patients, too, will experience various positive changes.
Benefits of IoMT for patients
The benefits of the Internet of Medical Things for patients are inseparable from the benefits for providers: healthier, better-managed patients cost less to treat, stay out of the hospital longer, and generate better outcomes data for value-based contracts. But the patient-side case stands on its own. Here is what IoMT delivers for the people on the receiving end of care.
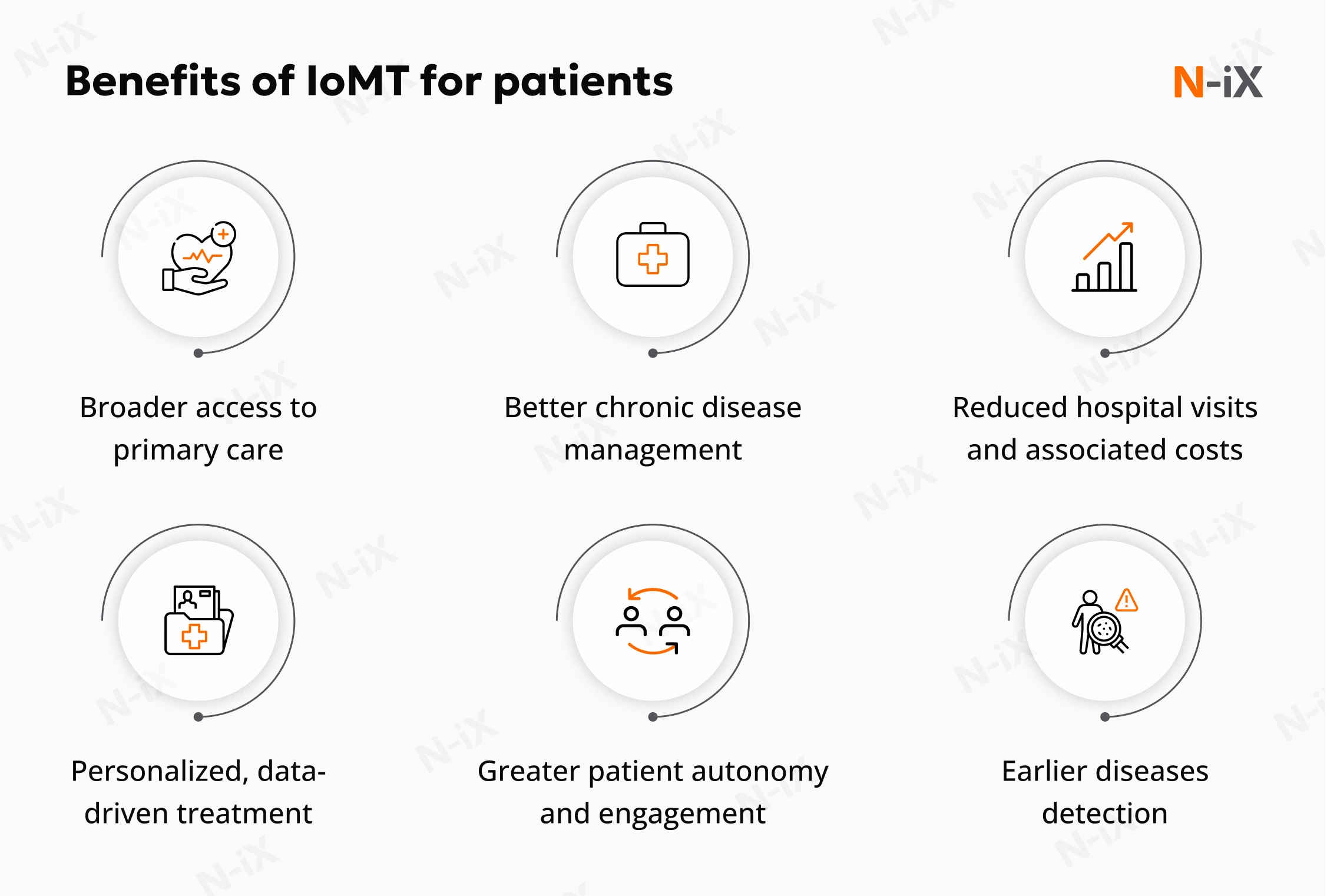
Broader access to primary care
IoMT-powered point-of-care kiosks enable residents in remote areas to access primary care services. Patients in rural or underserved areas can complete a clinical consultation, receive a prescription, or be referred to a specialist without having to travel to a facility.
This matters financially as well as clinically. Earlier access to primary care reduces the likelihood that conditions will escalate to emergency or specialist-level treatment, where healthcare costs rise, and patient outcomes deteriorate.
Better chronic disease management
For patients living with diabetes, hypertension, heart failure, or COPD, the traditional model of periodic clinic visits is inadequate as conditions shift daily. This is where IoMT-powered monitoring devices come into play.
Continuous glucose monitors, connected blood pressure cuffs, and cardiac wearables give patients and their care teams a real-time view of patient's conditions. This remote capability makes it convenient for patients to integrate at-home monitoring into their daily routines. For patients, this means fewer complications, fewer emergency episodes, and a better quality of daily life.
Personalized, data-driven treatment
IoMT provides physicians with a continuous stream of patient-specific data on how a patient actually responds to a medication, how their vitals shift with activity or stress, and when early signs of intolerance appear.
That data enables earlier intervention, faster dose adjustment, and reduced trial-and-error in treatment decisions. Patients spend less time on ineffective medications and experience fewer avoidable side effects. In conditions where treatment response varies significantly between individuals, as it does in oncology, autoimmune disease, and complex cardiac cases, this personalization is clinically significant.
Reduced hospital visits and associated costs
Every avoided hospital visit is a direct financial saving for the patient. It translates into fewer co-payments, lower transport costs, and less time off work. For patients managing chronic conditions that previously required monthly or weekly check-ins, the cumulative savings are substantial.
Beyond convenience, chronic disease self-management programs (CDSMPs) supported by remote monitoring tools have demonstrated average savings of $700 per person in emergency room visits and hospitalizations [8].
Greater patient autonomy and engagement
Patients with visibility into their own health data engage more actively with their treatment. They can see the direct impact of medication adherence, lifestyle changes, or missed doses, and that feedback loop drives behavior in ways that clinical instruction alone typically does not.
When patients are active participants in managing their conditions, adherence improves, outcomes improve, and the overall cost of care decreases.
Earlier diseases detection
IoMT shifts the clinical model from reactive to proactive. Wearables and connected sensors monitor continuously, not episodically. Anomalies such as an irregular cardiac rhythm, a sustained blood pressure spike, a pattern suggesting early-stage deterioration are flagged before they produce symptoms severe enough to require an emergency visit.
While there are plenty of advantages to adopting IoMT solutions for healthcare businesses, there are also challenges.
Key challenges in implementing the Internet of Medical Things
IoMT comes with unique legal, regulatory, technical, and privacy challenges because the IoMT ecosystem is complex and has many stakeholders, including:
- Medical device manufacturers;
- Connectivity providers
- Original equipment manufacturers (OEM);
- Systems/software engineers;
- System integrators;
- End users.
That's why Internet of Medical Things solutions should consider the requirements pertaining to each stakeholder. However, there are a few common IoMT challenges concerning each group.

Device integration
Many medical devices come from different manufacturers and use varying data formats and communication protocols. The lack of standardization makes it difficult for devices to communicate seamlessly with each other and with existing hospital IT systems, such as electronic health records (EHRs) and medical records (EMRs).
Compatibility issues between new IoMT devices and legacy systems can require significant investment in software development and IT support to enable proper integration. The issue becomes even more prominent if you want to scale such solutions. These challenges can increase both the time and costs associated with adopting IoMT, making seamless device integration a critical hurdle for healthcare providers.
N-iX solution: Adopt interoperability standards such as HL7, FHIR, or DICOM to ensure that IoMT devices and systems can communicate using a common language. Additionally, partnering with software development companies experienced in IoMT integration can help design custom solutions that ensure seamless integration of new devices with healthcare systems.
Explore our comprehensive guide to EMR & EHR software development
Data interoperability
Even when devices are technically connected, the data they produce is often incompatible. Different formats, inconsistent taxonomies, and fragmented pipelines make aggregating and analyzing multi-device data a significant processing effort. It, in turn, introduces opportunities for error.
For clinical decision-making that depends on complete, real-time data, the format of the data matters. Delayed or inconsistent data can slow care and undermine the value of the IoMT investment.
N-iX solution: The mentioned communication standards (HL7, FHIR, DICOM) also ensure data interoperability. Use integration platforms or middleware that bridge various devices and IT systems, simplifying data exchange. You can also choose cloud-based interoperability platforms to centralize data from various IoMT devices, streamlining access to and analysis of information from different sources in a unified manner.
Data security and privacy
The IoMT ecosystem usually includes numerous devices that are potential targets for cybercriminals. As the IBM Cost of a Data Breach Report 2025 states, data breach costs in 2025 remain the highest in the healthcare sector, reaching up to $7.42M [9].
Attackers now use AI to accelerate phishing and credential attacks. Shadow IoT, or unmanaged devices connected to hospital networks without IT visibility, has become an entry vector. A breach no longer just exposes data; it can disable the clinical systems patients depend on.
Since healthcare ranks among the top five industries most affected by cybercrime, businesses adopting the Internet of Medical Things should implement robust cybersecurity practices and policies.
N-iX solution: To prevent breaches, each IoMT device and its connected systems should transfer and collect data in accordance with specific security protocols. To respect patient privacy, the business should also implement data minimization (i.e., collecting only strictly necessary data) and pseudonymization (i.e., substituting real data identifiers with pseudonyms). In addition, if the business outsources a part of medical device software development, it should ensure its tech partner follows development protocols to avoid data breaches. Outsourcing partners should also have reliable fallback procedures if the devices' functionality is compromised.
Regulatory compliance and standards
Healthcare is one of the most regulated industries. Usually, the greater the risks a device or tech solution can pose, the stricter the regulations governing its production or development.
Moreover, each country has its own regulations governing data collection and processing by medical devices and healthcare solutions. For example, here are the similarities and differences in data requirements across the US, the UK, Canada, Australia, the UAE, and Qatar.
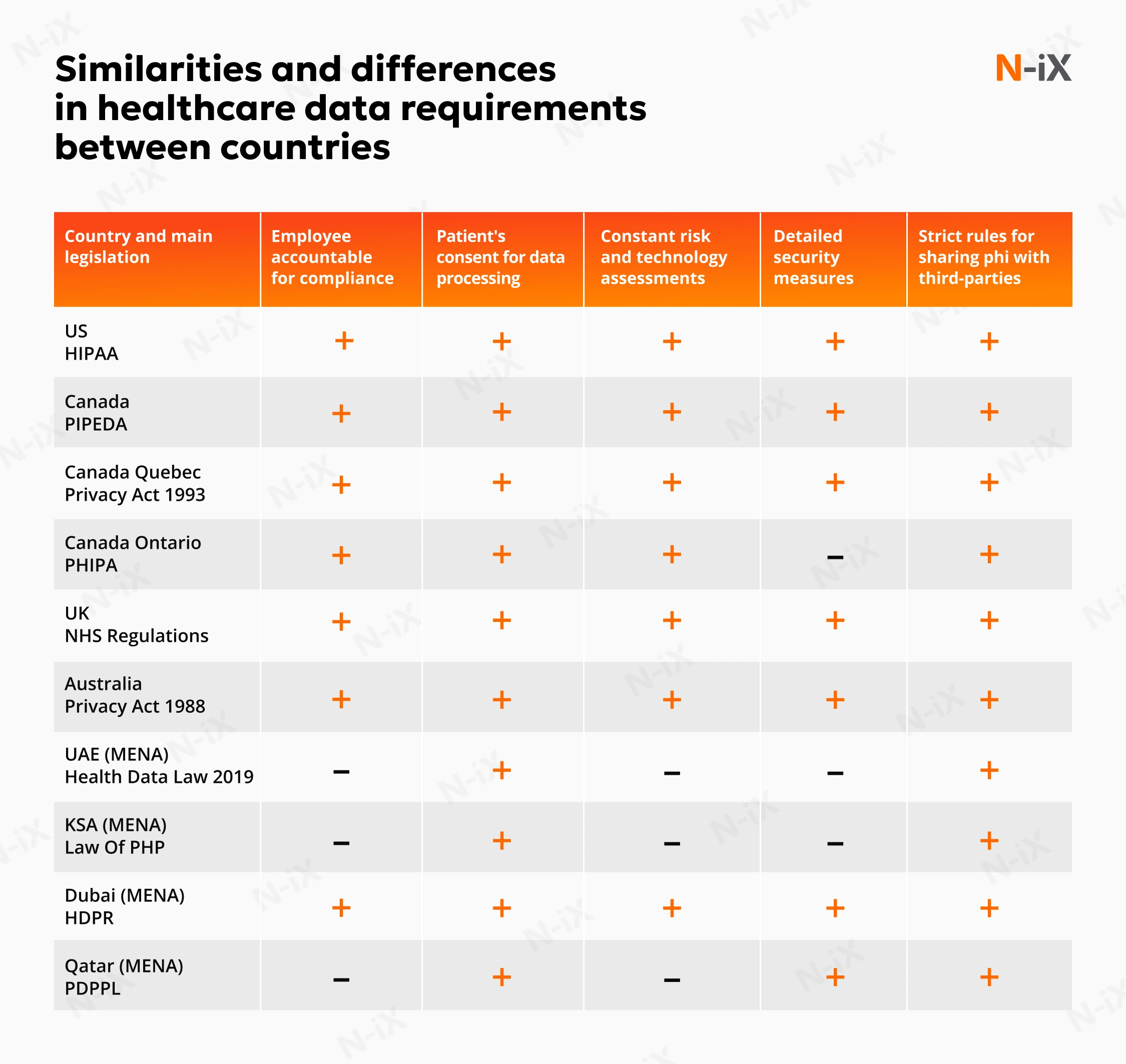
The European Union's regulations shaping IoMT user security are classified into separate categories. They include:
- Strictly “consumer” regulations: Directive 2011/83/EU;
- Safety regulations: General Product Safety Regulation, Medical Device Regulation (MDR);
- Cybersecurity regulations: NIS Directive, Cybersecurity Act;
- Provisions on the protection of personal data: General Data Protection Regulation (GDPR).
For organizations operating in the EU, the picture has grown significantly more complex since 2024. In addition to the existing MDR, GDPR, NIS Directive, and Cybersecurity Act, two major regulatory instruments now apply directly to IoMT:
- EU Cyber Resilience Act (CRA). Entered into force in December 2024, with reporting obligations applying from September 2026. It imposes mandatory cybersecurity requirements on all connected digital products, including IoMT devices, throughout their entire lifecycle [10].
- EU AI Act. It’s planned to be fully applicable as of August 2026, with additional obligations for high-risk AI systems, which include AI-enabled medical devices subject to MDR, taking effect in August 2027. Organizations deploying AI-powered IoMT in the EU must now address transparency, human oversight, documentation of accuracy, and cybersecurity resilience requirements that go beyond MDR alone [11].
However, you should consider the regulations governing the use, manufacturing, software development, and deployment of Internet of Medical Things devices within the target market. Here’s a list of major documents currently in force on the biggest healthcare markets:
- United States: HIPAA, HITECH;
- Canada: CMDR, PIPEDA;
- Brazil: RDC 751/2022;
- Japan: PMD Act;
- China: MDSАР;
- India: MDR 2017;
- Australia: TGA1989, MDR 2002.
The list includes the major documents, but depending on the device, software, or intended use, you may also need to follow additional requirements. Moreover, you should comply with international standards such as ISO or IEC. If you outsource any part of IoMT software or device development, adherence to the requirements, methodologies, and processes mentioned will help you pass audits and obtain the necessary certifications.
N-iX solution: Our compliance includes PCI DSS and SOC-2, ensuring we meet the strictest data security requirements. Additionally, we hold ISO 27001 for information security management, ISO/IEC 27701:2019 for privacy information management, and ISO 9001 for quality management systems. We’re also GDPR-compliant, making us a reliable tech partner for European healthcare businesses and minimizing data breaches and non-compliance risks.
Explore more: Achieve quality and compliance with a healthcare app development company
How N-iX can help businesses with IoMT
Bringing an Internet of Medical Things solution to market involves more than building a device or deploying software. It requires navigating clinical requirements, regulatory frameworks, legacy infrastructure, and cybersecurity obligations simultaneously. Here are a few examples of how the N-iX team can help you.
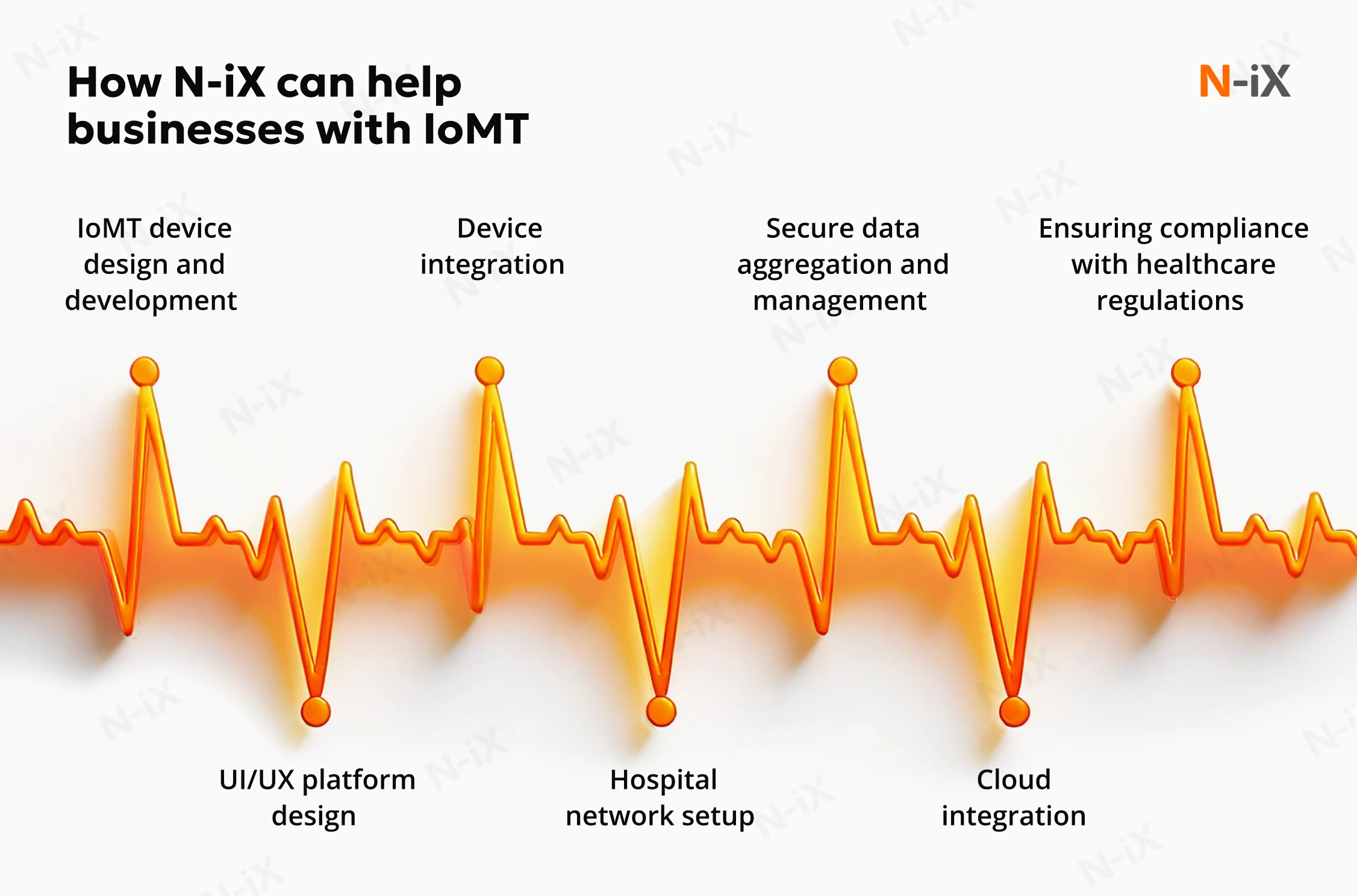
IoMT device design and development
N-iX offers comprehensive design and development services for IoMT devices, focusing on designing reliable, user-friendly, and scalable medical devices and robust software. We examine the existing IoMT ecosystem, study the physical setting, and plan for solution integration or the development of a standalone IoMT tool. It helps us create a solution or design that fits the intended setting and meets operational and business goals.
Our engineers combine embedded systems and IoT expertise with hands-on knowledge of medical device and firmware standards. Every device we build is designed to meet both technical performance requirements and clinical validation criteria.
For an Australian healthcare manufacturer, N-iX built the embedded firmware and remote management application for an air decontamination system used in clinical environments to prevent the spread of airborne infections. We delivered a stable, maintainable platform and a mobile interface that gave facility staff full remote control over the system. The result was a more reliable, easier-to-manage solution ready for deployment across healthcare facilities.
UI/UX platform design
Clinical data is only valuable if the people using it can act on it quickly. Our UI/UX team designs intuitive, user-centric interfaces for IoMT applications and platforms, including dashboards, patient portals, and clinician tools to present complex data clearly, minimize cognitive load, and support multiple user types within the same system. This expertise extends to telemedicine software development, where the quality of the interface directly determines whether a remote clinical interaction is as effective as an in-person one.
We have experience designing an innovative healthcare platform for Cure Forward, a leading digital platform that educates cancer patients about therapies and clinical trials in the US.
The N-iX team delivered a consistent, intuitive UI for the application that enables users to quickly access the data they need while supporting interactions among multiple users, optimizing efficiency, and minimizing response times. Additionally, we enhanced the patient portal with a system that automatically adjusts based on the user's responses. It helped us deliver a tailored experience to each user.
Device integration
N-iX helps seamlessly integrate IoMT devices into existing healthcare IT systems, including Electronic Health Records, Electronic Medical Records, and Hospital Information Systems (HIS). We address interoperability and standardization challenges, ensuring that devices communicate effectively to create a cohesive ecosystem.
Hospital network setup
We design and implement robust network infrastructure to support IoMT devices, ensuring reliable, secure connectivity (Wi-Fi, Bluetooth, etc.) across hospital environments. N-iX optimizes network performance to accommodate the high data traffic generated by connected medical devices, helping achieve real-time data transmission. Our solutions ensure low-latency, high-speed IoT connectivity, critical for continuous remote patient monitoring and effective healthcare delivery.
Secure data aggregation and management
Our data scientists ensure that data from multiple IoMT devices is consolidated smoothly, creating a unified view of patient data. We implement advanced encryption protocols, access control measures, and data validation processes to maintain data integrity and prevent unauthorized access. In addition, we offer end-to-end IoT security services for your IoT systems and develop solutions for every layer to ensure long-term resilience.
Cloud integration
We facilitate cloud integration for IoMT solutions, enabling scalable data storage and processing. N-iX's partnerships with AWS, Microsoft Azure, and Google Cloud Platform ensure seamless integration with leading cloud platforms. Our expertise in cloud architecture enables medical organizations to leverage real-time analytics and Machine Learning to derive actionable insights from IoMT data.
Ensuring compliance with healthcare regulations
N-iX helps businesses navigate the complex landscape of regulations, including GDPR, MDR, and ISO, while our knowledge of HIPAA requirements enables us to address broader regulatory needs.
Our team ensures that IoMT solutions are designed and implemented in compliance with the necessary standards, reducing the risk of data breaches and related legal issues. We conduct compliance assessments and security audits to ensure that your IoMT solutions meet the required standards of data privacy and patient safety.
For example, we helped our client, WEINMANN Emergency, to expand its development capabilities. Together with our client's team, our embedded and IoT experts have worked on developing compliant firmware for a new version of MEDUCORE Standard², a portable defibrillator. As a result, our client's product has met various regulatory requirements (Medical Devices Directive 93/42/EEC, EN 1789, EN 60601-2-27, EN 80601-2-61, RTCA DO-160, MIL-STD 810 G, EUROCAE ED-14G, etc.) and was cleared to be used within the EU.
Explore further: Healthcare IT outsourcing: Top factors to consider
Conclusion
The Internet of Medical Things offers immense potential for transforming healthcare through connected devices that enable better patient care, improved operational efficiency, and real-time data insights. Providers using IoMT-powered remote monitoring are cutting hospital admissions. Organizations with continuous patient data are winning value-based contracts. And as the global IoMT market expands, the gap between early adopters and those still evaluating is widening each year.
At the same time, the barriers are real. They include device integration across legacy infrastructure, data interoperability at scale, a cybersecurity threat landscape, and a regulatory environment that added the EU AI Act and Cyber Resilience Act to an already complex compliance stack. These challenges require technical depth, healthcare domain knowledge, and experience delivering solutions that meet clinical and regulatory standards simultaneously.
That is where N-iX comes in. With 23 years of experience and a team of over 2,400 software experts specializing in IoT, embedded systems, cloud, AI, and data science, we have the expertise to take IoMT projects from concept to certification-ready deployment.
We have worked with over 160 companies, including Fortune 500 firms and healthcare businesses like WEINMANN Emergency and Cure Forward. If your organization is working on an IoMT project and needs an experienced tech partner, get in touch. We will help you move faster, with a solution built to last and fewer compliance surprises.
Sources:
- Internet of Medical Things Market Growth Driven by Rising Adoption of Connected Healthcare Devices | Precedence Research
- FreeStyle Libre | Abbott
- Portable Single-Lead Heart Monitor | KardiaMobile | Kardia
- Telemedicine Kiosks for Healthcare | Telehealth | REDYREF
- 2025 Tech Trends Report | Future Today Strategy Group (FTSG)
- BlueSync™ Technology for Cardiac Rhythm | Medtronic
- The rise of robotics and AI-assisted surgery in modern healthcare | PMC
- Chronic Disease Self-Management Education Programs | ACL Administration for Community Living
- Cost of a Data Breach Report 2025 | IBM
- Regulation 2024/2847 Cyber Resilience Act CRA | Official Journal of the European Union
- Regulation 2024/1689 Artificial Intelligence Act | Official Journal of the European Union
FAQ
What is the difference between IoT and IoMT?
IoT refers to any connected device that exchanges data over a network, from smart thermostats to logistics sensors. IoMT is a specialized subset focused exclusively on healthcare. The key distinctions are purpose, regulation, and stakes. IoMT devices handle protected health information (PHI), must comply with regulations such as HIPAA, GDPR, and the EU Medical Device Regulation, and are subject to clinical validation requirements that consumer IoT devices are not.
What are examples of IoMT devices?
IoMT devices span five operational environments. Wearables include medical-grade continuous glucose monitors such as FreeStyle Libre and cardiac monitors such as KardiaMobile. Mobile IoMT includes smartphone-paired diagnostic tools. Public IoMT includes telehealth kiosks deployed in community settings. In-hospital IoMT includes sensor-equipped beds, connected infusion pumps, and real-time asset-tracking systems. Connected implants and AI-assisted surgical tools represent the frontier of the category.
How does IoMT support value-based care?
Value-based care reimburses providers based on patient outcomes and documented proof, not service volume. IoMT is the infrastructure that enables outcome-based care to operate at scale. Connected devices provide continuous, documented evidence of patient health status, such as glucose trends, cardiac rhythms, and blood pressure patterns. It supports quality reporting, reduces avoidable readmissions, and demonstrates the clinical results that payers reward.
What are the biggest challenges in implementing IoMT?
The most common implementation challenges are device integration, data interoperability, cybersecurity, and regulatory compliance.
How is AI changing the Internet of Medical Things?
AI helps shift IoMT from reactive monitoring to predictive care. Connected devices feed continuous data into models that flag deterioration risks and forecast readmissions before clinical signs appear. In addition, AI can enhance surgery, as AI-assisted connected robotic systems have shown a reduction in operative time and fewer intraoperative complications. At the same time, AI becomes a tool for cybercriminals, leading to a significant increase in cyberthreats across the industry.
What is the difference between IoMT and telehealth?
Telehealth is a care delivery model that includes remote consultations, digital prescriptions, and virtual visits. IoMT is the hardware and data infrastructure that makes telehealth clinically meaningful.
Have a question?
Speak to an expert





